CASE REPORT | https://doi.org/10.5005/jp-journals-10079-1051 |
Thoracic Fungal Osteomyelitis with Compressive Myelopathy in an Immunocompetent Individual by a Rare Fusarium Species: A Case Report
1–6Department of Orthopaedics and Spine Surgery, Preethi Institute of Medical Sciences and Research, Preethi Hospitals (P) Ltd, Madurai, Tamil Nadu, India
Corresponding Author: Vinoth Thangamani, Department of Orthopaedics and Spine Surgery, Preethi Institute of Medical Sciences and Research, Preethi Hospitals (P) Ltd, Madurai, Tamil Nadu, India, Phone: +91 7418804447, e-mail: drvinoththangamani@gmail.com
How to cite this article Sivakumar R, Muthu C, Nagendra Reddy SKV, et al. Thoracic Fungal Osteomyelitis with Compressive Myelopathy in an Immunocompetent Individual by a Rare Fusarium Species: A Case Report. TNOA J Orth Joint Surg 2021;3(2):91–94.
Source of support: Nil
Conflict of interest: None
ABSTRACT
Background: Thoracic vertebral osteomyelitis/spondylodiscitis and spinal epidural abscess are rare but life-threatening conditions, commonly seen in immunocompromised persons. It requires early detection and prompt management, to avoid late sequelae and complications. The cause for this abscess can be pyogenic, tuberculosis, fungal, or parasitic. Candida and Aspergillus species are the most common fungal causes which mainly present as discitis or osteomyelitis in the immunocompromised patient, after hematogenous dissemination.
Case discussion: We report about Fusarium, an opportunistic human pathogen which is a normal commensal of human skin flora, in a 60-year-old healthy gentleman with no immunocompromised stage, the rare cause of thoracic vertebral osteomyelitis with epidural abscess D9 to D11 with compressive myelopathy and neurological deficit in this article.
Conclusion: Along with aerobic, anaerobic bacterial and tuberculous, microbiological and histopathological investigations should also include fungal workup. Proper fungal isolation from the tissue is a must for prompt treatment and a better outcome for the patient.
Keywords: Dorsal spine epidural abscess, Fungal osteomyelitis, Fusarium.
INTRODUCTION
Fungal spondylodiscitis/osteomyelitis first described by Keating in 19321 can be caused by a wide spectrum of organisms like pyogenic, tuberculosis, fungal, or parasites. The peak incidence is at a mean age of 50 years2 in immunosuppressed individuals, requiring early detection and prompt management. The rate of occurrence of fungal infections has increased over the last decade among immunocompromised patients with a reported incidence of fungal spondylodiscitis is about 5% most commonly by species such as Candida and Aspergillus.3Fusarium is an emerging human opportunistic pathogen that is a normal commensal of human skin flora, lives in soil and water, and also causes mycotic infection in plants.4 Since the presentation of fungal osteomyelitis is vague, months would have been delayed before the initial presentation.
Here, we report our case of a dorsal spine vertebral osteomyelitis with epidural abscess causing compressive myelopathy due to rare Fusarium species in an immunocompetent individual, which in literature search shows no similar case.
CASE DESCRIPTION
Immunocompetent heavy labor of 60 years age male who had trivial work-spot fall and injured to his upper back (D10 D11 region) 2 years ago, for which he was treated as back muscle strain. Since the patient does not have any relief of pain, he took ayurvedic oil massage (2 episodes a week apart) to his back in the 3rd month of injury. But still continued to have dull aching back pain and ultimately became non-ambulatory about 5 months before presenting to our hospital.
On examination, there were no constitutional symptoms. His bowel and bladder functions are normal. Clinical examination revealed tenderness over D9–D12 region with normal truncal skin. Neurological examination of the spine was significant with paraplegia of the lower extremities (Frankel B grade) with associated thoracic compressive myelopathy features. Routine laboratory investigations were within the normal limits—except C-reactive protein level (CRP) of 55 mg/L (range 0.3–8), and an erythrocyte sedimentation rate (ESR) of 73 mm/hour (range 0–30) on the higher side. Imaging of the thoracolumbosacral spine (X-ray, CT, and MRI) showed brilliantly enhancing epidural lesion at D9–D10 region with cord compressive myelomalacia and an erosive lesion in the D10 right pedicle suggestive of osteomyelitis and D10–D11 epidural granulation tissue compressing the cord (Figs 1 to 3).
Surgical Intervention
Since the patient had a worsening neurological deficit, he was planned for surgical debridement, decompression (D10, D11 laminectomy), and pedicle screw stabilization (D9 to D11). Intraoperative gross examination of the lesion revealed a single piece-meal of cherry red soft-tissue mass, compressing on the cord and removed in toto of about 2 cc volume (Fig. 4). Samples (debris and black granules with epidural abscess) were sent for aerobic/anaerobic, tuberculosis and fungal culture, Gene Xpert, and histopathology. Postoperatively, the patient was put on an intravenous antibiotic as per our hospital protocol. TB-PCR was negative.
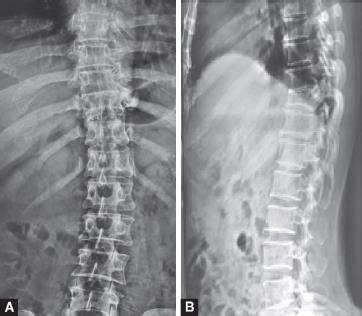
Figs 1A and B: Preoperative X-ray shows D10 right pedicle destructive lesion with D9–D10 decreased disk space and D10 superior end-plate collapse
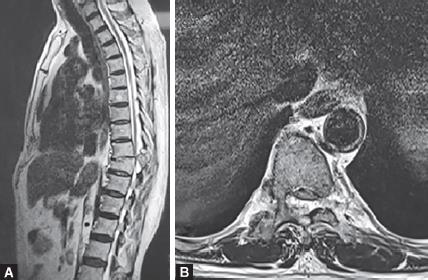
Figs 2A and B: T2-weighed image shows right D10 pedicle destruction and cord compression by epidural mass

Figs 3A and B: Contrast MRI shows brilliantly enhancing epidural abscess compressing the cord with the right irregular pedicle
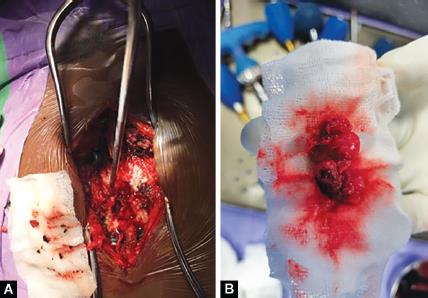
Figs 4A and B: Intraoperative—blackish fungal element along with reddish granulation tissue and D10 right pedicle excised in toto
Follow-up
For the first 2 weeks, all the cultures showed no growth, and in extended culture cottony hyaline filamentous fungal growth with hyphae branching at 45° was seen and it was found to be the rare fungal Fusarium species. Histopathology sections studied from received material showed large areas of hemorrhages, granulation tissue infiltrated by a mixed inflammatory infiltrate, necrosis with multiple fragmented viable and degenerated fungal hyphae invading the tissue. Periodic acid-Schiff (PAS) stain confirmed the fungal hyphae. Immediately after the fungal report around the third postoperative week, we started him on intravenous liposomal amphotericin B (5 mg/kg/day) for 2 weeks followed by oral voriconazole 200 mg twice daily dosage for 8 weeks. The patient showed remarkable neurological improvement from Frankel B to D at the end of 4 months of treatment. 1-year follow-up X-ray/MRI/CT scan (Figs 5 and 6) showed a good radiological D10 pedicle healed lesion seen with no signs of recurrence. Functional OSWESTRY disability index score decreased significantly from 71.1% preoperatively to 24.4% postoperatively, with complete neurological recovery.
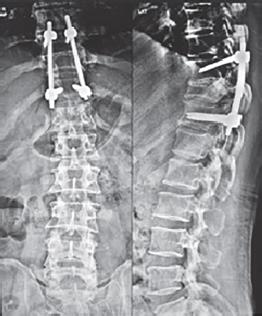
Fig. 5: Postoperative X-ray shows D9–D11 posterior stabilization with D10 and D11 post-laminectomy status
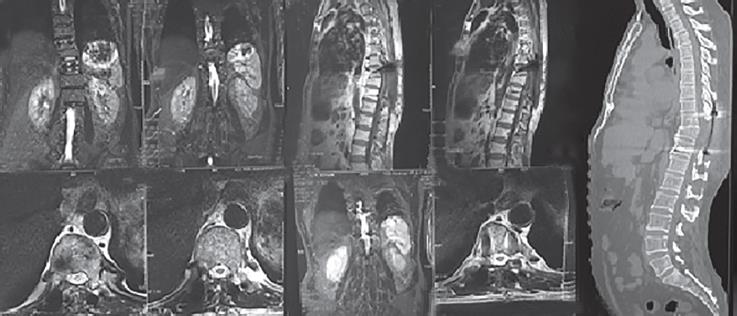
Fig. 6: One-year postoperative MRI and CT scan shows healed D10 right pedicle and no evidence of recurrent cord compression
DISCUSSION
Vertebral osteomyelitis with spinal epidural abscess, although rare, is still considered a serious condition that may lead to neurological complications. Dull aching low back pain for >1 month is a very common presenting complaint in 83% of patients.
Fungal pathogens reach the spine via hematogenous spread or by direct external inoculation/local invasive or by spread from contiguous tissues.5 Direct inoculation can be by a breach in skin most commonly iatrogenic, following spinal surgery, epidural procedures, lumbar puncture, and accounts for up to 25–30% of cases in some spondylodiscitis series.
Blunt trauma may result in hematoma formation before the symptoms of spinal epidural abscess arise in 15–35% of cases6 which subsequently becomes infected.7 In our patient, a trauma that resulted in the form of oil massage in the thoracolumbar paraspinal area (D9–D11) was the most likely route of inoculation; hence, it is important to consider fungal epidural abscess in patients even without evidence of immunosuppression, as colonization and tissue break down is the leading risk factor for infection.8
Violaceous nodule or papule with black eschar in the center surrounded by gray halo typical of cutaneous fusariosis at the site of entry is not found in our case.
A spinal epidural abscess will have 44% of coexistent vertebral osteomyelitis9 as our patient has the colonization of a fungus in the pedicle of the D10 vertebra.
Ninety-five percent of the cases of fungal spondylodiscitis involve the lower dorsal and lumbar spine similar to tuberculosis. Fungal spondylodiscitis also spares the intervertebral disk, a predilection for the anterior vertebral body, and paraspinal abscess formation may overlap with findings on the MRI scan. CT scan shows bone punctuated holes with a small island of irregular bone similar to TB.2 MRI with contrast is ideal for early diagnosis through HPE confirmation of the organism is the gold standard.10
Clinical Summary of Musculoskeletal Fusarium Cases Reported (Table 1)
Fusarium spp. are usually susceptible to amphotericin B and generally exhibit high MICs to echinocandins, flucytosine, and azoles, although they may exhibit intermediate susceptibility to voriconazole and posaconazole. The host immune system is the most important factor predicting outcome.8 Based on the data available, we recommend voriconazole and lipid-based amphotericin B formulations.
| Author/year | Region | Immune status |
|---|---|---|
| Jakle et al./1983 | Joint | Immunocompromised |
| Srilatha Edupuganti et al./2011 | Vertebral abscess and osteomyelitis | Immunocompromised |
| Asmita Sagar Sakle/2012 | Peritoneum | Immunocompromised |
| Bourguignon et al./1976 | Lower extremity (tibia) | Immunocompetent |
| Page et al./1982 | Lower extremity (4th toe) | Immunocompetent |
| Nuovo et al./1988 | Lower extremity (leg) | Immunocompetent |
| Sierra-Hoffman et al./2005 | Osteomyelitis | Immunocompetent |
| Our case | Spine lower thoracic | Immunocompetent |
Multidisciplinary approach10 in form of surgical debridement, antifungals in the advice of infectious control specialist for prescribed time and rehabilitation with braces prevents relapse and complications.
Prompt diagnosis of fungal infections depends on the pathologist’s skill and adequacy of the organism received in the biopsy. Fusarium oxysporum can be cultured in non-cycloheximide11 Sabouraud dextrose agar with chloramphenicol to suppress secondary bacterial growth.12 A polymerase chain reaction followed by DNA sequencing allows early identification of Fusarium. Fusarium is best visualized in tissue stained with Grocott silver and less well with hematoxylin-eosin stain sections.5
Though literature advises antifungals for 6–12 months duration, we limit our patient to 10 weeks since surgical debrima eradicated the foci.13 High chance of negative histopathology in a single sample and hence the current recommendation is to obtain a minimum of six samples from different parts of the lesion.
CONCLUSION
To our knowledge, this is the first case report of epidural abscess due to rare Fusarium spp. The diagnosis and treatment of fungal infection of the spine are often delayed, as clinical suspicion is often low, particularly in immunocompetent individuals. Preventing fusariosis relies on detection and treatment of cutaneous damage before commencing immunosuppression and decreasing environmental exposure to Fusarium.
REFERENCES
1. Keating P. Fungus infection of bone and joint. South Med J 1932;25(10):1072–1079. DOI: 10.1097/00007611-193210000-00016.
2. Kim CW, Perry A, Currier B, et al. Fungal infections of the spine. Clin Orthop Relat Res 2006;444:92–99. DOI: 10.1097/01.blo.0000203451.36522.4c.
3. Titlic M, Josipovic-Jelic Z. Spondylodiscitis. Bratisl Lek Listy 2008;109(8):345–347.
4. Tupaki-Sreepurna A, Kindo AJ. Fusarium: The versatile pathogen. Indian J Med Microbiol 2018;36(1):8–17. DOI: 10.4103/ijmm.IJMM_16_24.
5. Gupta AK, Baran R, Summerbell RC. Fusarium infection of the shin. Curr Opin Infect Dis 2000;13(2):121–128. DOI: 10.1097/00001432-200004000-00005.
6. Verner EF, Musher DM. Spinal epidural abscess. Symposium on infections of the central nervous system. Med Clin North Am 1985;69(2):375–384. DOI: 10.1016/s0025-7125(16)31049-5.
7. North JB, Brophy BP. Epidural abscess: a hazard of spinal epidural anaesthesis. Aust NZ J Surg 1979;49(4):484–485. DOI: 10.1111/j.1445-2197.1979.tb05847.x.
8. Nucci M, Anaissie E. Cutaneous infection by fusarium species in healthy and immunocompromised hosts: implications for diagnosis and management. Clin. Infect. Dis. 2002;35(8):909–920. DOI: 10.1086/342328.
9. Darouiche RO, Hamill RJ, Greenberg SB, et al. Bacterial spinal epidural abscess: review of 43 cases and literature survey. Medicine 1992;71(6):369–385. DOI: 10.1097/00005792-199211000-00004.
10. Caldera G, Cahueque M, Cobar A, et al. Fungal spondylodiscitis: review. J Spine 2016;5(02):2. DOI: 10.4172/2165-7939.1000302.
11. Romano C, Caposciutti P, Ghilardi A, et al. A case of primary localised cutaneous infection due to fusarium oxysporum. Mycopathologia 2010;170(1):39–46. DOI: 10.1007/s11046-010-9290-9.
12. Sreedharan Namboothiri PE, Nair SN, Vijayan K, et al. Disseminated fusarium oxysporum neurospinal infection. Indian J Orthop 2014;48(2):220–222. DOI: 10.4103/0019-5413.128773.
13. Yang SC, Fu TS, Chen LH, et al. Identifying pathogens of spondylodiscitis: percutaneous endoscopy or CT-guided biopsy. Clin Orthop Relat Res 2008;466(12):3086–3092. DOI: 10.1007/s11999-008-0441-y.
________________________
© The Author(s). 2021 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and non-commercial reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.